A great example of this is a leaf that is dry and curled up. Most people would look at such a leaf and conclude that the plant was getting insufficient water, and that the plant owner is implicitly at fault for not watering the plant enough. Or, people may blame the weather or climate, saying it was a dry spring, dry year, climate is changing, etc. All these diagnoses are based upon simple observation with little or no factual information backing them up.
Alternatively, some experts will admit that the symptoms are vague, and suggest sending a sample into a state diagnostic lab. The drawback is that this is a bit time consuming and expensive. Furthermore, these labs may not discover the root cause of the disease. That's because they chiefly identify a disease by trying to culture it. If they can't culture it, they can't identify it.
In the above "dry leaf" example, surprisingly, the available water may not be an issue at all. Rather, the plant leaves may be unable to get this available water due to their internal plumbing being choked with fungus.
One solution to this diagnosis problem is to examine the interior and exterior of plant tissue (twigs and leaves) using a hand-held computer-based USB microscope. This type of analysis complements both a visual analysis and plant tissue culture analysis, since plant tissue damage at the microscopic level can be very revealing. And sometimes it alone is sufficient to diagnose the responsible pathogen.
Still, I've found that even having lots of simple microscopic photos isn't as helpful as it could be, since each photo, due to hardware limitations, is a relatively tiny view of the plant being examined. I'm reminded of the story of the group of blind men trying to describe an elephant by examining different parts of it. Each man will report different characteristics, making for a confusing description. Only a photo of the entire animal will make things really clear. Similarly, where an individual microscopic photo is limited in usefulness, a coherent collection of them is far helpful. Hence, I've developed a way to combine up to 200 individual microscopic photos into one panoramic view. I chiefly use this technique to examine the cross-section of a diseased twig.
To illustrate this, see photos 1 through 4, which are individual microscope photos from a cross-section of a diseased Colorado blue spruce twig.

|

|
|

|
|||
| Photo 1 - Blue spruce cross-section 1 | Photo 2 - Blue spruce cross-section 2 | Photo 3 - Blue spruce cross-section 3 | Photo 4 - Blue spruce cross-section 4 |
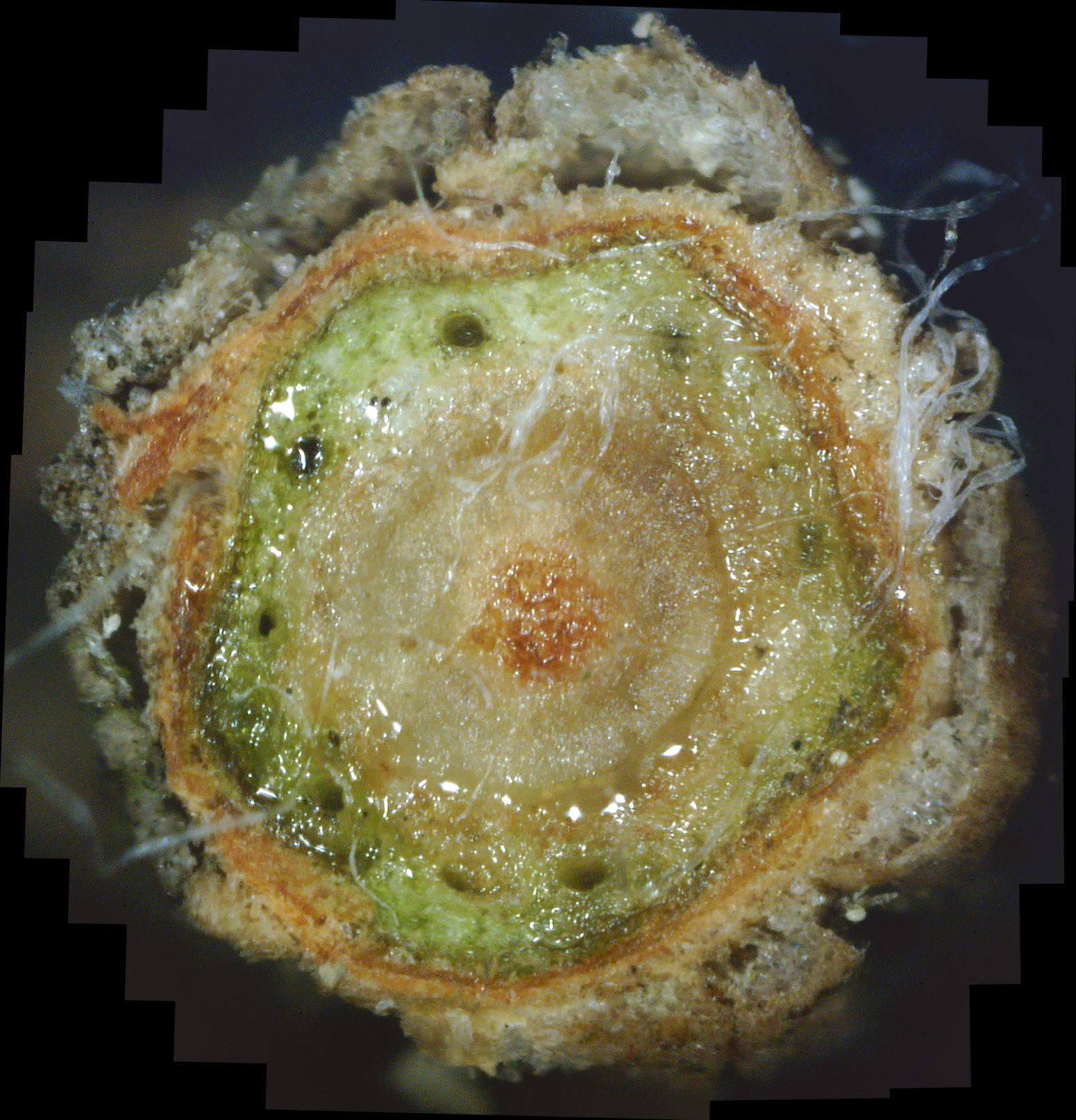
While these views are interesting, they fail to give a clear picture of what is going on inside this twig. While only 4 photos are shown above, in actuality 47 of these photos were gathered. But trying to display all 47 would be overwhelming due to the mental effort required to assimilate them into a whole. Instead, if these individual photos properly overlap, computer software can be used to "stitch" them together into a single large panorama view, as shown in photo 5 below.
|
| Photo 5 - Colorado blue spruce cross-section panorama |
Furthermore, no resolution is lost, as a click on the composite photo will bring it up in "full screen" view, and another click will cause the view to zoom in even further.
To one degree or another, all Colorado blue spruce trees in our area seem to be fighting a white canker infection. Some trees are doing better than others. Photo A1 shows a twig cross-section from a blue spruce that is only moderately infected. Photo A2 shows a twig cross-section from a dead branch on blue spruce (the tree was still alive, though).

|

|
|
| Photo A1 - Blue spruce cross-section 1 | Photo A2 - Blue spruce cross-section 2 |
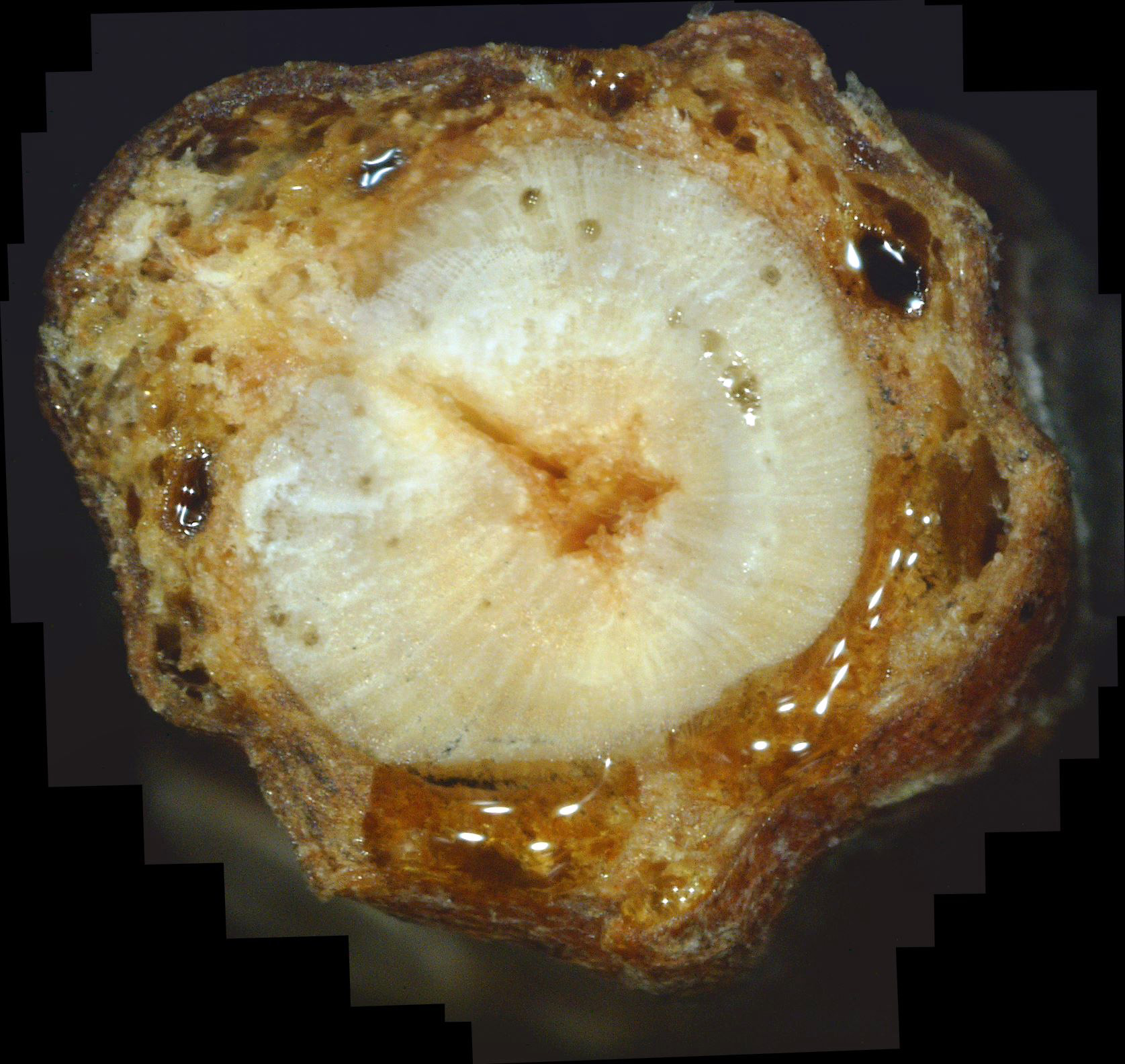
My neighbor has a large white pine that has been in gradual decline for years. One day I noticed that a dead branch had dropped from it into my yard. So I decided to do a cross-section of the branch. The result is shown in photo A3. Once again, a mass of white canker fungus appears to have choked this branch to death.
|
| Photo A3 - Dead white pine twig cross-section |
When a twig dies, the healthy, cholorphyl tissue typically shrinks, while the white canker tissue does not. Also, careful examination will show that white canker growth tends to avoid the sap producing vacuoles.
California
Since I enjoy gardening, a few months ago I became aware of a newly introduced beautiful
dogwood tree called an Aka Tsuki. Unfortunately, since it was summer, all local
sources of this tree had been sold out. In desperation, I found a source in California, and
ordered it from a commercial grower there. I wondered if it, too, might have a white canker
infection. So, within an hour of arrival at my home, I unpacked it, took a twig from it, made a
cross-section of it, and scanned this cross-section with my microscope. I did this within
an hour since I didn't want to chance infecting this new tree with white canker from our
area. The cross-section is shown in photo B1.

|
| Photo B1 - Aka Tsuki dogwood twig cross-section |
As you can see, this twig is full of white canker, as is the bark surface! In retrospect, that wasn't a big surprise, since when I opened the shipping container, the leaves looked somewhat wrinkled and limp. But without knowing more, one could easily assume that was due to the stress of shipping. Oh, I should also add that the tree was well packed and the dirt around the roots was moist, so this wasn't a shipping problem.
In spite of a recommendation to let this tree "sit" in its container throughout the summer (to let its root system mature), I planted it immediately and gave it a soaking of both water and fungicide propiconazole. Months later, this tree has not declined nor grown.
So, even trees originating in California, 3,000 miles away, can have a white canker infection.
Florida
In late spring we made a visit to relatives living in West Palm Beach, Florida.
Since I have a strong interest in both trees and this white canker disease, I decided to
check out their property for some interesting trees I had seen in past visits over the
years. The two trees I examined were called Gumbo Limbo trees. They have unusual,
smooth peeling bark and only grow in the southern Florida area. But during this visit, I
noticed that these trees looked like they were in decline: branches were dying and the
leaves on them looked somewhat sick. So I clipped off a branch and brought it back home
with me to check it out. I should add that they had a commercial tree care company examine
this tree, and he could not identify the cause of the decline.
At the time, I wasn't heavily into panoramic twig cross-sections, so I just took a number of non-overlapping microscope photos of a cross-section of this sample branch. The results are shown in photos B2 through B5.

|

|
|

|
|||
| Photo B2 - gumbo limbo cross-section 1 | Photo B3 - gumbo limbo cross-section 2 | Photo B4 - gumbo limbo cross-section 3 | Photo B5 - gumbo limbo cross-section 4 |
Once again, the characteristics of white canker popped up. Recalling my visit there last year, even then I noticed a slight decline in these trees. But at the time I didn't pay much attention to it since the decline wasn't as noticeable as now, and I didn't think white canker could be that widespread.
So, apparently white canker disease occurs across the entire country. In a sense, that's not so strange, since every tree and shrub I've tested seems to have some degree of infection by white canker. Some plant pathologists find this too astounding to be true. In fact, I'd have to agree, were it not for the huge amount of photographic evidence I've accumulated.
Cosmos
Both my wife and I like cosmos plants. They have nice delicate leaves, beautiful flowers, and
bloom for a long time in summer. However, this summer (2014), we noticed that some of the
cosmos plants looked unhealthy. My wife attributed it to a lapse in our garden watering
efforts. However, I wondered if white canker was involved. So, I cut a stem at two locations
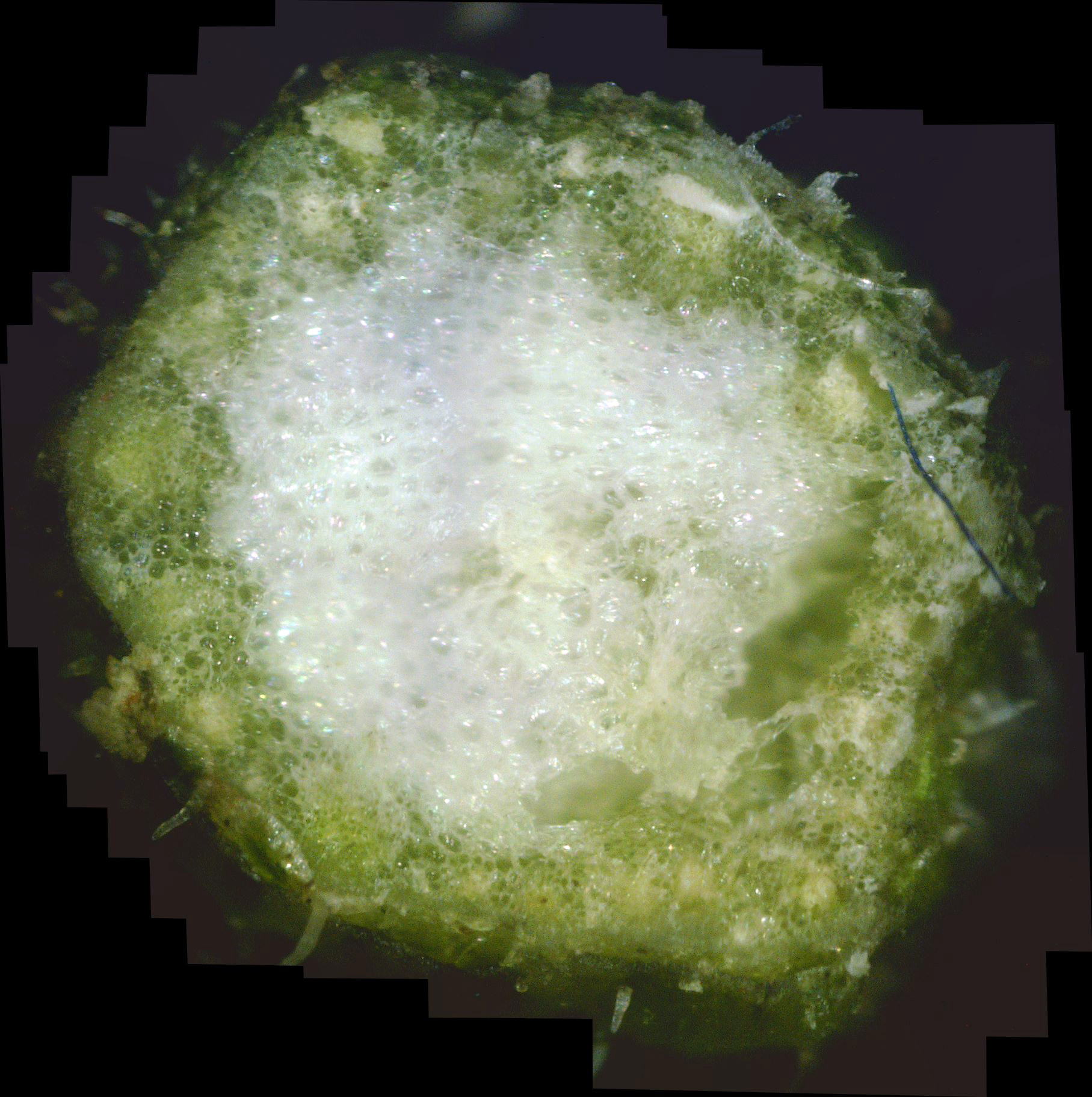
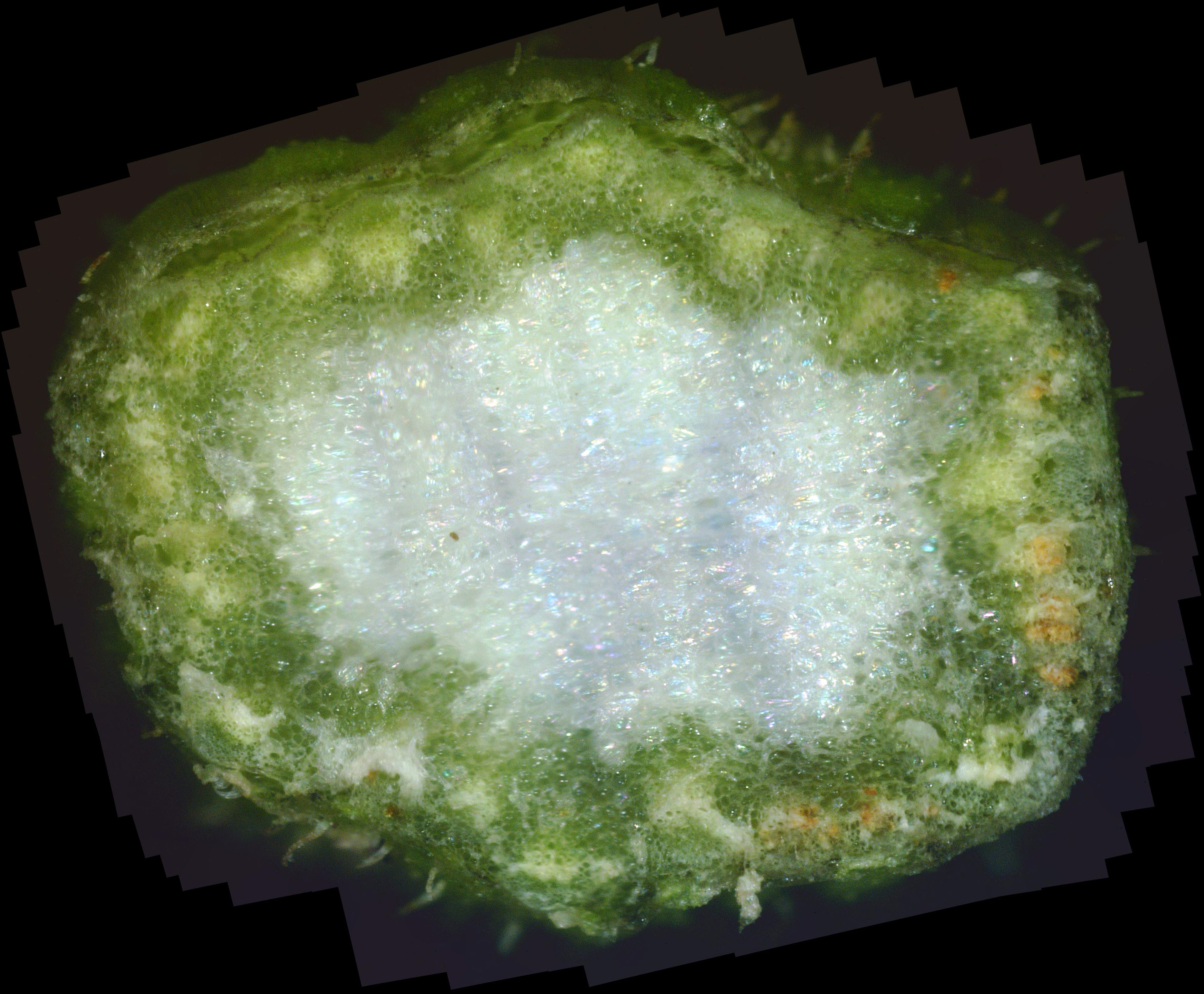
and made a cross-section near each cutting. Photos C1 and C2 show the results.
|
|
|
| Photo C1 - Cosmos stem cross-section 1 | Photo C2 - Cosmos stem cross-section 2 |
The stem shown in photo C1 is the smaller of the two, being higher up on the plant. At first glance, the mostly white pith and mostly green phloem look normal. But clicking on each photo to zoom into its interior shows that there is lots of "white stuff" between the green cells of the plant. This white material is likely white canker which is choking off the transport of water and nutrients to the plant. That may be the very reason why the plant looks like it hasn't been watered enough. So, my wife may be half-right in that the plant isn't getting enough water.
White Pine
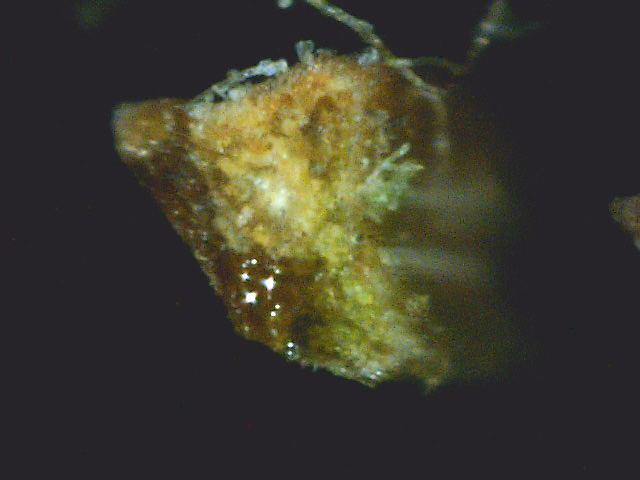
My neighbor has a white pine tree that seems a bit sick since it is continually losing
needles. Consequently, when the wind broke a small dead branch off and it fell on my property,
I decided to do a cross-section on its needles. The result is shown in photos D1 through D4.

|
|

|
||
| Photo D1 - White pine needle cross-section 1 | Photo D2 - White pine needle cross-section 2 | Photo D3 - White pine needle cross-section 3 |
Blue Spruce
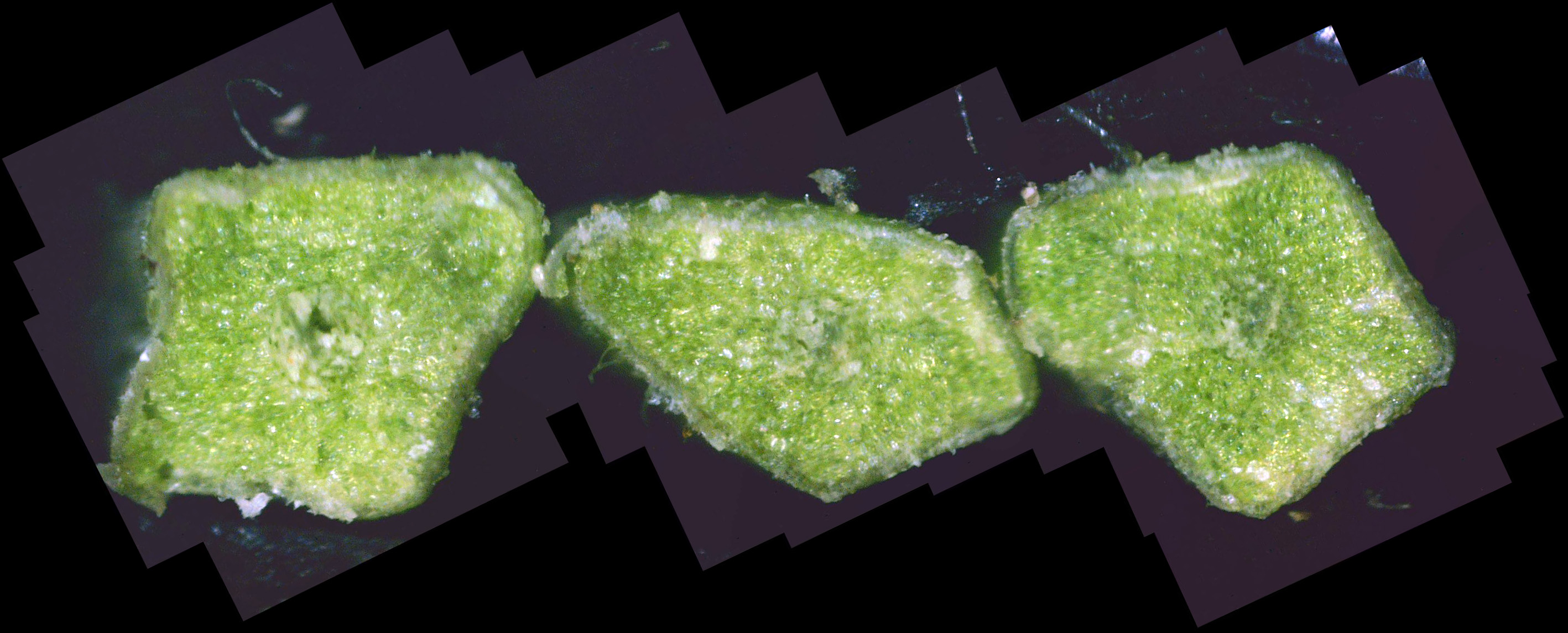
There is a mature Colorado blue spruce in our neighborhood that has been in gradual decline
for about six years. I thought it might be interesting to do a cross-section on some of its
needles. The result is shown in photo D4.
|
| Photo D4 - Blue spruce needle cross-section |
The needles seem relatively healthy, with only a smattering of white canker. Here's a case where a twig cross-section is more informative than a needle cross-section.
Fraser Fir
There is a small (about 5' high) Fraser fir in my yard that appears healthy, so I wondered
what a cross-section of one of its needles would look like like.
The result is shown in photo D5.

|
| Photo D5 - Fraser fir needle cross-section |
The interior is a nice rich green with no trace of blobs of white canker. So here we have a healthy evergreen needle for comparison. Compare it to the white-green interior of the diseased blue spruce above.